Cervical Cancer And HPV Testing
GenPath Women’s Health offers a number of different tests appropriate to assist in the diagnosis of cervical cancer, as well as HPV, as determined by the American College of Obstetricians and Gynecologists (ACOG).
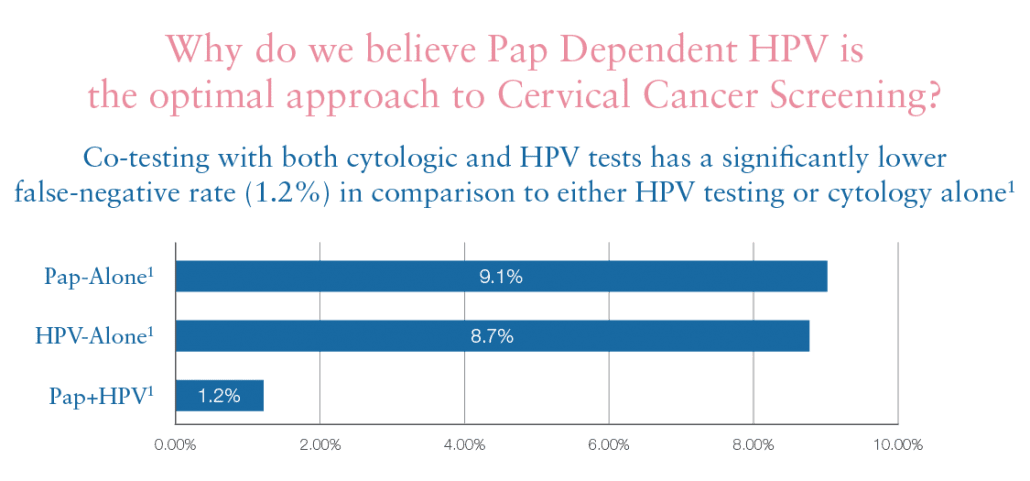
Pap Dependent HPV
Pap dependent HPV is GenPath’s unique approach to cervical cancer screening. It maximizes the opportunity for detection of patients at risk of cervical disease by selecting the best-suited HPV test based on a patient’s Pap results. HPV DNA levels are likely to be high in the initial phases of HPV infection and begin to decrease as the infection progresses, just as HPV mRNA levels begin to increase. GenPath matches an HPV DNA-based test to low-grade cytology classifications and an HPV mRNA-based test to higher-grade cytology classifications.
Cytology
GenPath utilizes the most advanced Pap test technology with capabilities in both the Hologic ThinPrep Imaging System with Dual Review and the BD FocalPoint™ Slide Profiler Intelligent Pap Imaging™.
Pathology
Our experienced, board-certified medical pathology experts review all results. Our pathologists employ the most current adjunctive tools, in conjunction with traditional technologies, to provide the most accurate diagnosis. Consultative services with our staff are available for your convenience.

